
His most well-known thought experiment involved photographing an electron. The Uncertainty principle is also called the Heisenberg uncertainty principle. This equation applies to complex molecules and to internal molecular energy levels as well. Heisenberg sometimes explained the uncertainty principle as a problem of making measurements. 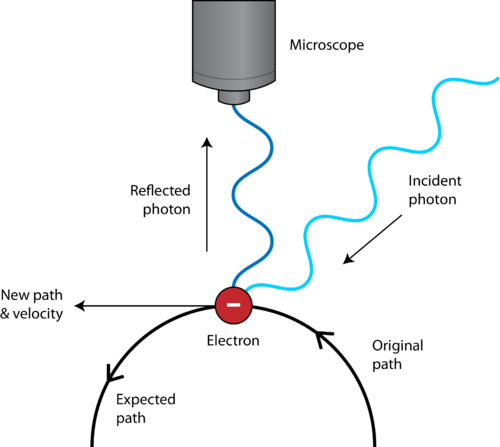
Thus, this principle is regarded as a fundamental principle of nature. There is likewise a minimum for the product of the uncertainties of the energy and time. There is a minimum for the product of the uncertainties of these two measurements. (c) the necessity of using all of them at different moments for a comprehensive account of the totality of phenomena that we must consider. (b) the possibility of applying each one of them separately at any given point and. Trying to pin a thing down to one definite position will make its momentum less well pinned down, and vice-versa. The position and momentum of a particle cannot be simultaneously measured with arbitrarily high precision. (a) a mutual exclusivity of certain phenomena, entities, or conceptions and yet. Heisenberg uncertainty principle synonyms, Heisenberg uncertainty principle pronunciation, Heisenberg uncertainty principle translation, English dictionary definition of Heisenberg uncertainty principle. A principle in quantum mechanics holding that greater accuracy of measurement for one observable quantity entails less accuracy of. It tells us that there is a fuzziness in nature, a fundamental limit to what we can know about. Heisenbergs uncertainty principle synonyms, Heisenbergs uncertainty principle pronunciation, Heisenbergs uncertainty principle translation, English dictionary definition of Heisenbergs uncertainty principle. Werner Heisenberg stumbled on a secret of the universe: Nothing has a definite position, a definite trajectory, or a definite momentum. T he uncertainty principle is one of the most famous (and probably misunderstood) ideas in physics. (ii) States: that if the time for which the system remains in a particular energy state is short, then its energy will be more defined and for a longer stay in a state, the energy will not be well defined. The Uncertainty principle is also called the Heisenberg uncertainty principle. Where ΔE and Δt are uncertainties in the energy of the particle and time of passage past a particular point. Though widely repeated in textbooks, this physical argument is now known to be fundamentally misleading.$$ \Delta E\times \Delta t\ge \frac $$ ………… (ii) This ascribes the uncertainty in the measurable quantities to the jolt-like disturbance triggered by the act of observation. uncertainty principle, also called Heisenberg uncertainty principle or indeterminacy principle, statement, articulated (1927) by the German physicist Werner Heisenberg, that the position and the velocity of an object cannot both be measured exactly, at the same time, even in theory.  
Later, in 1927, a German physicist called Werner Heisenberg proposed that it is not possible to know where exactly an electron is located when it behaves like a wave and what its velocity is simultaneously because by trying to take any measurements, we would be disturbing it in some way. The original heuristic argument that such a limit should exist was given by Heisenberg, after whom it is sometimes named the Heisenberg principle. Heisenberg Uncertainty Principle Definition. The formal inequality relating the standard deviation of position σx and the standard deviation of momentum σp was derived by Earle Hesse Kennard later that year and by Hermann Weyl in 1928. For instance, in 1927, Werner Heisenberg stated that the more precisely the position of some particle is determined, the less precisely its momentum can be known, and vice versa. Listen to Heisnbergs early thoughts on the uncertainty. In a nutshell: Dresden Codaks cartoon 'Lil Werner' Heisenbergs conclusions on the impact on physics. An account of a supporting thought experiment. Meaning of Heisenberg uncertainty principle. That is, the more exactly the position is determined, the less known the momentum, and vice versa. Uncertainty relations expressed mathematically. Definition of Heisenberg uncertainty principle in the dictionary. The Heisenberg uncertainty principle states that it is impossible to know simultaneously the exact position and momentum of a particle. Uncertainty principle In quantum mechanics, the uncertainty principle is any of a variety of mathematical inequalities asserting a fundamental limit to the precision with which certain pairs of physical properties of a particle known as complementary variables, such as position x and momentum p, can be known simultaneously. This page focuses on the origins of Heisenbergs uncertainty relations and principle.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
